The Pharmaceutical Supervision and Administration Bureau (ISAF) announced in a statement on Monday that a prescription medication from the United States used to treat blood cancer, “Tazverik Tablets 200mg” is being recalled, according to a notification from the local pharmaceutical importer.
The statement pointed out that the medicine manufactured by US-based “Patheon Pharmaceuticals Inc.”, is being voluntarily recalled by its producer.
The recall is based on ongoing clinical trial data which has indicated a potential increased risk of secondary haematological malignancies*, the statement said, adding that as this potential risk may outweigh the therapeutic benefits of the treatment, the manufacturer has initiated a voluntary recall of the product as a precautionary measure.
According to information provided by the importer, the affected medication was supplied to a number of private hospitals and polyclinics in Macau.
The bureau has instructed the importer to follow up on the recall. Medical institutions and clinics are directed to immediately stop using this medication, the statement added.
The bureau urged residents who have obtained the problematic medication from medical institutions or clinics to consult their attending physician or pharmacist for advice on how to proceed. For any enquiries, the public may contact the ISAF Monitoring Department on 8598 3233 during office hours.
* Haematological malignancies are a group of cancers that originate in the blood-forming tissues, such as the bone marrow, or in the cells of the immune system (lymphatic system). – DeepSeek
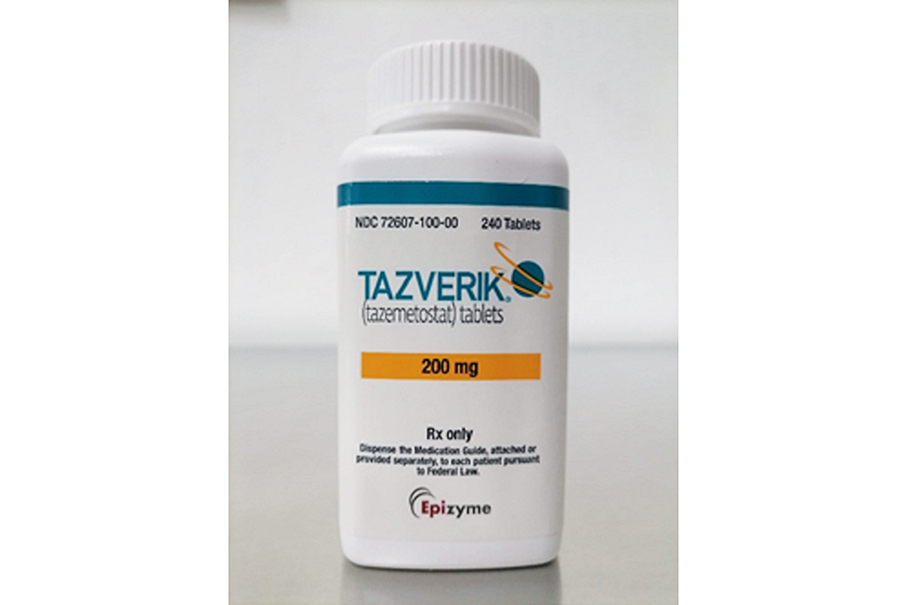
This undated handout photo provided by the Pharmaceutical Supervision and Administration Bureau (ISAF) on Monday shows the problematic “Tazverik Tablets 200mg”, produced by US-based “Patheon Pharmaceuticals Inc”.








